DECIPHERING THE TRICHOFERMA-PLANT DIALOG, IMPORTANCE OF ROOT EXUDATES
By: Sarai Esparza-Reynoso and José López-Bucio, Instituto de Investigaciones Químico-Biológicas, Universidad Michoacana de San Nicolás de Hidalgo, Morelia, Michoacán, México.
email: sariesparza@gmail.com; jbucio@umich.mx

Plants as primary producers of ecosystems host a myriad of microbial species, which rely on carbon-rich, root-exuded substances for their nutrition, including sugars, organic acids, and amino acids. A large part of the fungal and bacterial microbiome remains neutral, but a few species may establish pathogenic or symbiotic relationships that ultimately influence plant fitness, adaptation, and productivity (1). A major goal towards sustainable crop management is to identify and characterize microbial species with probiotic traits, from which it may be possible to identify highly active substances to develop the new generation of pesticides, biostimulants, and defense elicitors for the growing market. In this scenario, the fungal genus Trichoderma is attaining increasing importance since it comprises more than 200 reported species with versatile metabolism and well-adapted properties to proliferate in soil and water environments (2).
Research from the last two decades increased our understanding of the beneficial effects of Trichoderma to plants regarding 1) root branching and absorptive potential, 2) usage of organic amendments and fertilizers, 3) growth and development, and 4) adaptation to abiotic and biotic challenges. In summary, many species including T. virens, T. atroviride and T. longibrachiatum may help plants to survive better and enhance productivity in a safe and eco-friendly manner. The impact of these fungi has been assessed under field conditions, which correlated with yield increases in important cereal, fruit and vegetable crops, including maize, wheat, soybean, tomato, grape and lettuce (2).
In recent years, a major question has been how Trichoderma adjusts its metabolism according to the highly variable ecological niches and nutritional resources encountered. Apparently, secretion of potent enzymes such as cellulases, chitinases, and peptidases, are the hallmark that enables exploitation of dead wood and decaying leaf, root and stem materials and underscores its ability to parasitize phytopathogen fungi. However, it appears that the repression of genes encoding fungal degradative enzymes enables root colonization by Trichoderma and thus the fungus recognizes healthy roots by means of their exudation profiles (3).
Decoding the Trichoderma chemical message
Trichoderma is a biofactory of organic substances and releases volatiles, plant hormones, secondary metabolites and small peptides whose molecular composition depends on several factors including the fungal species, nutrient availability and the interaction with microorganism and plants. These info-chemicals can be perceived by roots through free diffusion within the soil and organic matter and during physical contact between the hyphae with the root epidermis, or at later stages, where the fungus spreads to inner cortical cells (4-6).
The first apparent change in the rhizosphere as a consequence of Trichoderma presence is the pH acidification (7). It may explain its highly efficient performance to solubilize sparingly soluble phosphates that accounts for a better plant nutrition. As the fungus grows, volatile emissions are thought to sensitize roots and enable long distance root-fungal recognition. 6-pentyl-2H-pyran-2-one (6-PP) is the main volatile from T. atroviride blends, which triggers root branching in Arabidopsis via changes in auxin and ethylene transport and response, respectively (4). 1-decene has been found to repress defense, stress and disease response genes, which facilitates fungal spread in root tissues (6). T. virens and T. asperellum releases auxins, a class of phytohormones with roles in plant growth and immunity that may be directly related to their biostimulant properties (8, 9).
Physical recognition may trigger further reactions in both the fungal and plant partners. Chitin, a major constituent of fungal cell walls has long been considered an elicitor that triggers defensive reactions in plants. Other molecules such as small peptides as well as membrane or cytoplasmic fungal proteins may further alert roots to be prepared for the interaction in order to avoid deleterious effects, making it much more competitive (10). Through the proliferation of lateral and adventitious roots, plants exploit better the mineral and water resources and are more resistant to abiotic stress, and these processes are effectively induced by Trichoderma.
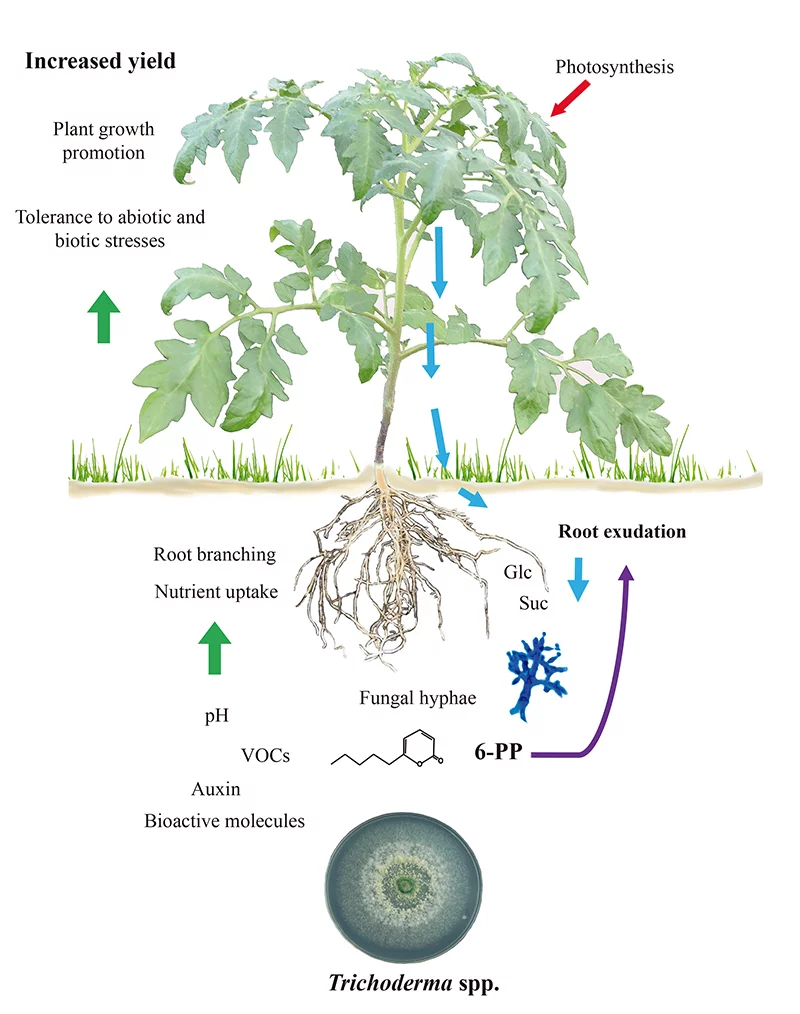
Figure 1.
Trichoderma-induced plant growth and defense depend on root exudates
Growth/defense tradeoffs in plants are modulated during biotic interactions. This ensures developmental transitions to flowering to proceed and warrant seed production. Since Trichoderma triggers both plant immunity and growth promotion, it is difficult to ascertain if these programs that are inherently costly may be linked. Moreover, it is possible that fungal-colonized roots could have an enhanced metabolism to support the energetic demand of the symbiosis. Carbohydrate exudation by roots from tomato seedlings has been found to increase in response to T. atroviride and sugars may act in a positive chemotactic response to attract the hyphae (11) as reported in figure 1. Consistently, the fungus expresses an intracellular invertase (Triat51014) and two putative sucrose transporters (Triat226844 and Triat83012) to use plant-derived carbon. On the other hand, T. virens takes up sucrose via a sucrose transporter and hydrolizes it through an intracellular invertase, which enables rapid growth by the energy provided by this disaccharide (12).
Molecular evidence helps explain how Trichoderma changes its metabolic signatures upon detection of root exudates. Villalobos-Escobedo et al. (2020) went further to demonstrate that the expression of genes encoding enzymes involved in complex carbohydrate degradation such as cellulose or chitin prior to root colonization is compromised in T. atroviride mutants defective on NADPH oxidase, an enzyme directly involved in the production of reactive oxygen species. Mutation of the corresponding genes not only affects lateral root formation and biomass production in the plant host, but also affects the defense reactions elicited and the fungal saprophytic behavior to acquire and use the simple sugars available in root exudates (3).
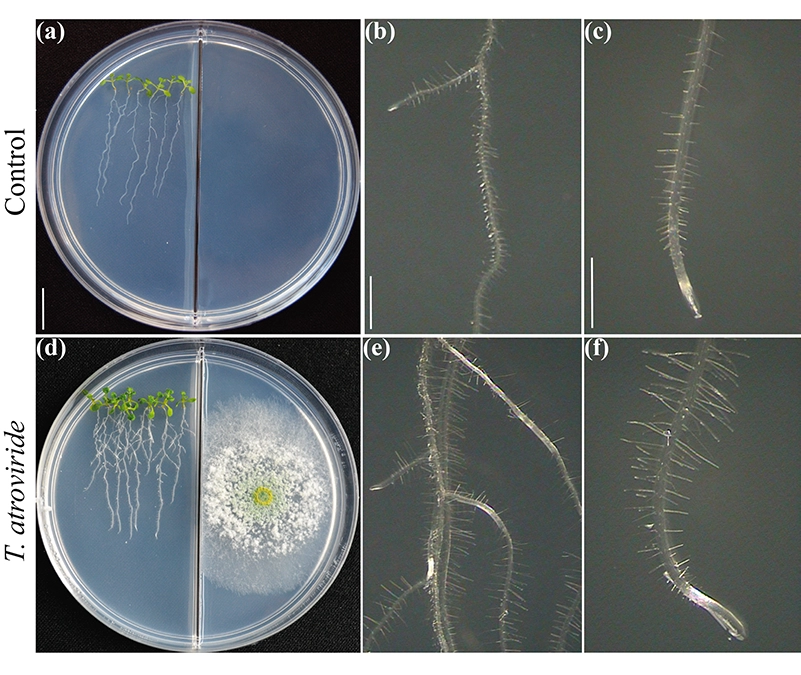
Figure 2.
It has been largely discussed whether the plant traits underlying high biomass production in interactions with microbes rely essentially on emitted carbon dioxide. Certainly, carbon dioxide is a major reactant for photosynthesis and should account for biomass enhancement. Nevertheless tight communication depends on the molecular composition of volatile blends. By testing plant growth and development in divided Petri plates to assess the fungal-plant interaction via volatiles (Fig. 2), we have unveiled the critical role of 6-PP for plant biomass production directly influencing hormonal responses, and shoot-root long distance transport of sugars (Esparza-Reynoso et al. submitted). It supports the notion that a regulatory loop for sugar distribution depending upon photosynthesis, the carbon status of the shoot and the perception of fungal metabolites are critical for mitosis in root meristems. Interestingly, plant growth promoting fungi including Trichoderma strains and Serendipita indica and S. williamsii strongly promote photosynthesis by means of no single compounds, but through mixtures of volatiles (13, 14). Feedback inhibition of carbon dioxide fixation due to elevated sugar levels apparently fails to occur in leaves exposed to fungal volatiles, possibly due to a hormonal imbalance. Plants are not alone, they rely on their fungal symbionts to survive and thrive. Time has come to translate the current knowledge into the implementation of Trichoderma for field applications.
- Cordovez V, Dini-Andreote F, Carrión VJ, Raaijmakers JM (2019). Ecology and evolution of plant microbiomes. Rev. Microbiol. 73:69-88.
- López-Bucio J, Pelagio-Flores R, Herrera-Estrella A (2015). Trichoderma as biostimulant: exploiting the multilevel properties of a plant beneficial fungus. Hort. 196:109-123.
- Villalobos‐Escobedo JM, Esparza‐Reynoso S, Pelagio‐Flores R, López‐Ramírez F, Ruiz‐Herrera LF, López‐Bucio J, Herrera‐Estrella A (2020). The fungal NADPH oxidase is an essential element for the molecular dialog between Trichoderma and Arabidopsis. Plant J. 103:2178-2192.
- Garnica-Vergara A, Barrera‐Ortiz S, Muñoz‐Parra E, Raya‐González J, Méndez‐Bravo A, Macías‐Rodríguez L, Ruiz‐Herrera LF, López‐Bucio J (2016). The volatile 6‐pentyl‐2H‐pyran‐2‐one from Trichoderma atroviride regulates Arabidopsis thaliana root morphogenesis via auxin signaling and ETHYLENE INSENSITIVE 2 functioning. New Phytol. 209:1496-1512.
- Lee S, Behringer G, Hung R, Bennett J (2019). Effects of fungal volatile organic compounds on Arabidopsis thaliana growth and gene expression. Fungal Ecol. 37:1-9.
- Guo Y, Ghirardo A, Weber B, Schnitzler JP, Benz JP, Rosenkranz M (2019). Trichoderma species differ in their volatile profiles and in antagonism toward ectomycorrhiza Laccaria bicolor. Microbiol. 10:891.
- Pelagio-Flores R, Esparza-Reynoso S, Garnica-Vergara A, López-Bucio J, Herrera-Estrella A (2017). Trichoderma-induced acidification is an early trigger for changes in Arabidopsis root growth and determines fungal phytostimulation. Plant Sci. 8:822.
- Contreras-Cornejo HA, Macías-Rodríguez L, Cortés-Penagos C, López-Bucio J (2009). Trichoderma virens, a plant beneficial fungus, enhances biomass production and promotes lateral root growth through an auxin-dependent mechanism in Arabidopsis. Plant Physiol. 149:1579-1592.
- Wang YF, Hou XY, Deng JJ, Yao ZH, Lyu MM, Zhang RS (2020). AUXIN RESPONSE FACTOR 1 acts as a positive regulator in the response of Poplar to Trichoderma asperellum inoculation in overexpressing plants. Plants (Basel) 9:272.
- Rocafort M, Fudal I, Mesarich CH (2020). Apoplastic effector proteins of plant-associated fungi and oomycetes. Opin. Plant Biol. 56:9-19.
- Macías-Rodríguez L, Guzmán-Gómez A, García-Juárez P, Contreras-Cornejo HA (2018). Trichoderma atroviride promotes tomato development and alters the root exudation of carbohydrates, which stimulates fungal growth and the biocontrol of the phytopathogen Phytophthora cinnamomi in a tripartite interaction system. FEMS Microbiol. Ecol. 94:fiy137.
- Vargas WA, Mandawe JC, Kenerley CM (2009). Plant-derived sucrose is a key element in the symbiotic association between Trichoderma virens and maize plants. Plant Physiol. 151:792-808.
- Harman GE, Doni F, Khadka RB, Uphoff N (2019). Endophytic strains of Trichoderma increase plants’ photosynthetic capability. Appl. Microbiol. https://doi.org/10.1111/jam.14368.
- Venneman J. et al. (2020). Respiratory CO2 combined with a blend of volatiles emitted by endophytic Serendipita strains strongly stimulate growth of Arabidopsis implicating auxin and cytokinin signaling. Plant Sci. https://doi.org/10.3389/fpls.2020.544435.



